 
Position and momentum are complementary properties of the electron. In short, you can see the electron as a particle or as a wave, but not both at the same time. You can’t see an electron as a particle and know its exact position, while you can’t know its precise velocity or momentum when it acts as a wave, at the same time. The natural duality of the electron is demonstrated by the de Brogile relation h being plank’s constant. The Electron on the Uncertainty PrincipleĪn electron is a particle, but it also behaves like a wave. Heisenberg and Pauli collaborated together in quantum mechanics. 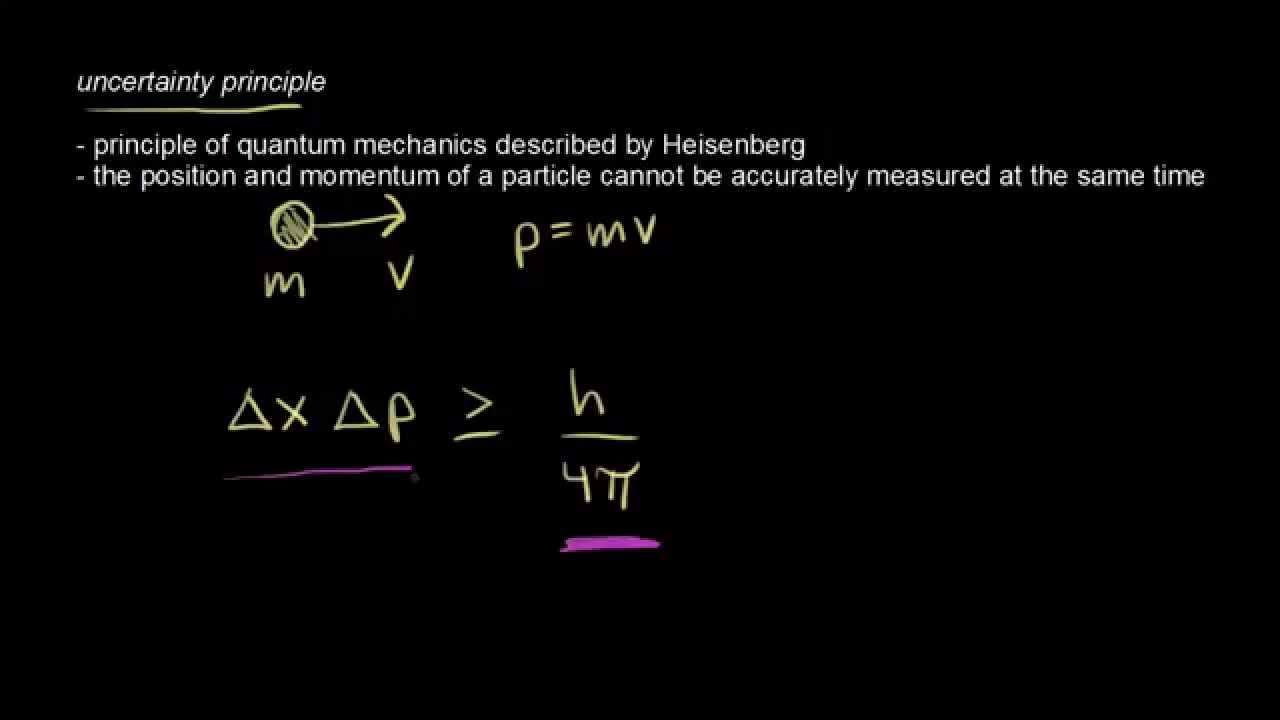
Pauli recommended that he dedicate himself to studying atomic theory, instead of the theory of relativity, since there was a greater field of study. Werner was the leader researcher on the atomic bomb project during World War II.ĭuring his doctorare years, he met Wolfgang Pauli. From 1924 to 1927 he got a scholarship to work with Niels Bohr at the Copenhagen University. Heisenberg was Max Borns assistant in 1923. He was born on December 5 th, 1901 in Wüzburgo and studied at the Munich University. He is best known for formulating the Uncertainty Principle, which was published in 1927, five years later he was awarded the Nobel Prize in physics for the “The creation of quantum mechanics, the use of which has led, among other things, to the discovery of allotropic forms of hydrogen.”. Werner Karl Heisenberg was a German theoretical physicist, who focused his studies on quantum mechanics. This principle tells us about limits and behaviors inside quantum mechanics. The Uncertainty Principle, formulated by Werner Heisenberg, states that we can’t know the exact position and momentum/velocity of a particle at the same time with perfect accuracy the more we know about the photon or electrons speed, the less we know about its position, and vice versa. After reading the article you’ll fully understand the electron duality and the basis of the principle Topics Covered in Other Articles In short: the uncertainty principle describes a trade-off between two complementary properties, such as speed and position.In this article you will learn about one of the fundamental principles on quantum mechanics, the uncertainty principle. Conversely, if we wanted to know the exact position of one peak of a wave, we would have to monitor just one small section of the wave and would lose information about its speed. 
The location is spread out among the peaks and troughs. The more peaks and troughs that pass by, the more accurately we would know the speed of a wave-but the less we would be able to say about its position. To measure its speed, we would monitor the passage of multiple peaks and troughs. To understand the general idea behind the uncertainty principle, think of a ripple in a pond. Quantum objects are special because they all exhibit wave-like properties by the very nature of quantum theory. 
Though the Heisenberg uncertainty principle is famously known in quantum physics, a similar uncertainty principle also applies to problems in pure math and classical physics-basically, any object with wave-like properties will be affected by this principle. In other words, if we could shrink a tortoise down to the size of an electron, we would only be able to precisely calculate its speed or its location, not both at the same time. Formulated by the German physicist and Nobel laureate Werner Heisenberg in 1927, the uncertainty principle states that we cannot know both the position and speed of a particle, such as a photon or electron, with perfect accuracy the more we nail down the particle's position, the less we know about its speed and vice versa. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
